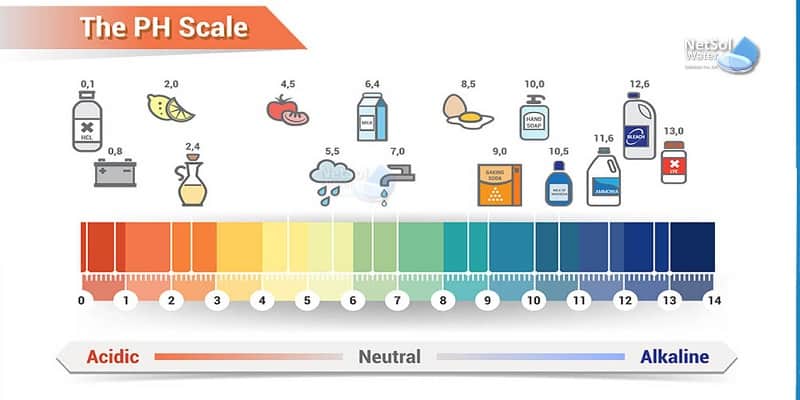
Alkalinity is one of the most important tasks to take place in a water or wastewater treatment plant. Also known as pH, and it is the measure of free hydrogen in water. Controlling the pH is important for every single process in a wastewater treatment plant to ensure the quality of the water. Read on to learn more about alkalinity adjustment in a wastewater treatment plant.
The Importance of pH in Wastewater Treatment
As mentioned, pH is used to control the quality of water produced in different steps in a water treatment plant. The target pH depends on the specific use for which the water being treated. This is true regardless of whether the treatment will be biological or chemical. To this end, a good pH regulation system is important if you want to get the right output.
This system generally comprises the
- Storage containers such as bulk bags, bulk silos, or bag hoppers in which the alkalinizing agent is stored.
- Screw feeders with shaftless screws that feed products accurately.
- An isolation injector which protects the screw feeder from humidity and moisture.
- Slurry preparation tank
- Packaged Pump skid for fluid handling needs
What Chemical is Used to Increase Alkalinity?
Due to the fact that different end-products require different degrees of alkalinity, there are many chemicals that can be used to achieve the right pH. Different chemicals also have different reaction rates, which affects the ability to control and adjust.
The main chemicals used to increase alkalinity in water are the four outlined below:
Hydrated or Quick Lime

It is also known as hydrated lime and quicklime. Since hydrated lime is alkaline, it is the most suitable chemical to treat acidic water. It increases the pH level and alkalinity of the water by neutralizing the agent. It softens hard water by removing minerals like magnesium and calcium making it potable. One of the major benefits is that it is more affordable than other chemicals used.
Caustic Soda

Caustic soda, commonly known as Sodium Hydroxide (NaOH), is available in a broad range of concentrations. It is a strong alkali which, means is can easily change the hard water pH to much closer approximation to neutral when injected into the water treatment system. At high concentrations, it’s dangerous to handle as it is highly soluble and must be used with a lot of precautions. These include having washing stations close by and using personal safety equipment for protection. Apart from this, caustic soda is also the most expensive chemical you can use for alkalinity adjustment.
Soda Ash

Soda ash or sodium carbonate is the third chemical that can be used to change alkalinity in water treatment plants. It is not as volatile as some of the other options, although the correct amount to be used depends on how much carbon dioxide is present in the water being treated. As such, it is usually added a little at a time until the desired alkalinity is achieved.
Magnesium hydroxide

Magnesium hydroxide is the powerful and cheapest alkaline chemical available in the market for pH control. It is produced by adding calcium hydroxide in seawater. It is self-buffering in nature as it will only solubilize to generate a pH of around 8.5 maximum. This means it is safe to handle, is non-toxic and non-corrosive.
Water treatment often requires rigorous pH control precision. pH regulation system should be a piece of technology that make sure that your water treatment methods are effective.
pH Regulation systems by Sodimate generally uses lime addition. The dry lime is stored on-site in a silo, hopper, or super sack that transferred to a lime slurry tank before feeding into the water. You may ask for a different setup depending on the final output you require. The team of professionals at Sodimate will work with you from the start to the end of your project, finding out exactly what you need for your plant and advising you accordingly. We deliver equipment made to the highest standard for your plant and in the specifications that you require.